Today, scientists are designing microscopic devices microbots and micromotors to eventually move through the body to perform medical tasks. Synthetic rods, tubes, helices, spheres or cages as small as a cell could be sent into the blood, liver, stomach or reproductive tract to diagnose conditions, carry drugs or perform surgery.
So far, most microbot experiments have been done in vitro under conditions very different from those in the human body. Many devices rely on toxic fuels, such as hydrogen peroxide. They are simple to steer in a Petri dish, but harder to control in biological fluids full of proteins and cells, and through the body's complex channels and cavities.
To enter clinical trials, microbots must clear two major hurdles. First, researchers need to be able to see and control them operating inside the body current imaging techniques have insufficient resolution and sensitivity. Second, the vehicles need to be biocompatible and be removed or stabilized after use.
https://giphy.com/gifs/l0Iy6gyl4L60K5lZK/html5
A magnetically driven helix captures an immotile bovine sperm and carries it to an egg cell.
M. Medina-Sánchez, L. Schwarz, A. K. Meyer, F. Hebenstreit & O. G. Schmidt Nano Lett. 16, 555561 (2016)
All three micromotor types share challenges. The materials they are made from must be proved to be biocompatible (such as polymers; metals including gold and zinc; proteins and DNA) or biodegradable (alginate, gelatin, calcium carbonate). They need to be able to perform a wide range of tasks: from sensing and responding to their environment to storing and delivering molecules or cells when stimulated by physical cues or by certain molecules, disease biomarkers, temperatures or levels of acidity. They need to be more manoeuvrable in three dimensions, in viscous and elastic body fluids and in phantom organs. And their targeting must be accurate.
Per chi volesse leggere tutto l'articolo: http://www.nature.com/news/medical-m...ontrol-1.22022
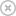



 Rispondi Citando
Rispondi Citando




